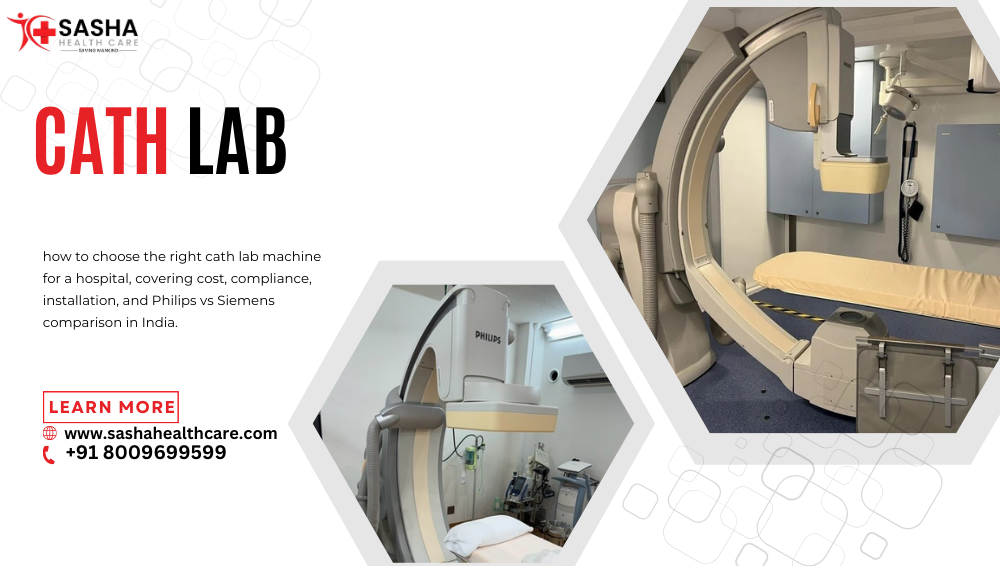
How to Choose the Right Cath Lab Machine for a Hospital
How to Choose the Right Cath Lab Machine for a Hospital? Selecting a Cath lab machine is a capital decision that impacts clinical outcomes, regulatory compliance, and long-term revenue. A cardiac catheterization laboratory (Cath Lab) is an advanced imaging suite used for minimally invasive cardiac and vascular procedures under fluoroscopic guidance. This guide provides a structured, evidence-based framework for hospital owners, radiologists, and healthcare decision-makers in India.
How to Choose the Right Cath Lab Machine for a Hospital
Choosing the right Cath lab machine requires a structured evaluation of clinical needs, infrastructure readiness, regulatory compliance, and long-term financial planning.
A Cath lab system must match the hospital’s intended procedure mix. Facilities focused on routine coronary angiography require different specifications than tertiary centers performing structural heart or neuro-interventions. Detector size, imaging software, radiation management features, and C-arm flexibility determine procedural efficiency.
Hospitals in India must ensure compliance with the Atomic Energy Regulatory Board before installation. Room shielding, power stability, and radiation safety protocols must align with national standards.
Leading manufacturers such as Philips and Siemens Healthineers offer modular Cath lab platforms designed for upgradeability and long-term scalability.
SASHA HEALTH CARE supports hospitals in technical comparison, site planning, vendor evaluation, and regulatory coordination. A structured assessment reduces risk, protects investment, and ensures the Cath lab supports both present clinical demand and future expansion.
What Is a Cath Lab Machine?
A Cath lab machine is a fixed angiography imaging system that produces real-time X-ray images during interventional cardiology and vascular procedures.
Core components include:
- Digital flat-panel detector
- High-frequency X-ray generator
- C-arm with motorized movement
- Patient table with weight-bearing capacity
- Hemodynamic monitoring system
- Image processing workstation
Modern systems support:
- Coronary angiography
- Angioplasty (PTCA)
- Structural heart interventions
- Peripheral vascular procedures
- Neuro-interventions (in biplane setups)
“Cath lab quality directly influences procedural precision, radiation exposure, and patient safety.”
Key Clinical Factors to Evaluate
1. Procedure Volume and Case Mix
Hospitals must first define:
- Expected daily caseload
- Type of interventions (diagnostic vs interventional)
- Adult vs pediatric cardiology
- Structural heart program plans
Single-plane systems suit routine coronary work.
Biplane systems suit neuro and structural heart programs.
High-volume tertiary centers require advanced image processing and workflow automation.
2. Detector Size and Image Quality
Flat-panel detector sizes:
- 20×20 cm (focused coronary work)
- 30×30 cm (multi-specialty use)
Larger detectors reduce repositioning and improve efficiency.
Image clarity depends on:
- Detector resolution
- Dose management algorithms
- Motion compensation technology
Lower radiation with preserved image quality defines a premium system.
Philips Cath Lab Systems
Philips manufactures advanced image-guided therapy platforms under the Azurion range.
Notable platform:
- Philips Azurion 7
Key features:
- FlexArm geometry
- ClarityIQ dose reduction
- Integrated hemodynamic monitoring
- Touchscreen workflow control
Typical price range in India:
₹1 crore – ₹8 crore (depending on configuration)
Philips systems suit high-end cardiac and hybrid Cath labs.
Siemens Cath Lab Systems
Siemens Healthineers develops angiography systems under the ARTIS platform.
Leading models:
- ARTIS pheno
- ARTIS icono
Key features:
- PURE image processing
- CARE radiation dose optimization
- Robotic C-arm movement
- 3D imaging capability
Typical price range in India:
₹1 crore – ₹7.5 crore
Siemens systems perform well in structural heart and complex vascular procedures.
Installation Requirements in India
Cath lab installation requires detailed infrastructure planning.
Room Specifications
- Minimum 800–1,000 sq ft
- Ceiling height: 3–3.5 meters
- Lead shielding as per AERB norms
- Controlled access entry
Power and Electrical
- Dedicated UPS
- Stable three-phase power
- Generator backup
Compliance and Licensing
Hospitals in India must obtain approval from:
- Atomic Energy Regulatory Board
Radiation safety officer appointment required.
“Cath lab approval without AERB clearance is not permitted in India.”
Budget Planning and Total Cost of Ownership
Capital cost alone does not define financial viability.
Consider:
- Annual Maintenance Contract (AMC)
- Comprehensive Maintenance Contract (CMC)
- Tube replacement cost
- Detector replacement cost
- Software upgrades
- Staff training
Annual maintenance costs:
5–10% of equipment cost.
Return on investment depends on:
- Procedure volume
- Case complexity
- Insurance tie-ups
Safety and Radiation Management
Radiation exposure affects:
- Patients
- Cardiologists
- Technologists
Advanced systems include:
- Real-time dose monitoring
- Automatic collimation
- Pulsed fluoroscopy
Hospitals must maintain:
- Personal dosimeters
- Lead aprons (0.5 mm equivalent)
- Radiation logbooks
“Dose optimization protects staff longevity and patient safety.”
Comparing Philips vs Siemens Cath Lab
| Parameter | Philips | Siemens |
|---|---|---|
| Workflow | Touchscreen-centric | Robotic integration |
| Dose Reduction | ClarityIQ | CARE + PURE |
| Structural Heart | Strong | Strong |
| Neuro Capability | Available (biplane) | Excellent (biplane) |
| Upgrade Path | Modular | Modular |
Both brands maintain global service networks and certified engineers in India.
Choice depends on hospital goals, clinical team preference, and long-term service support.
How SASHA HEALTH CARE Supports Hospitals
SASHA HEALTH CARE assists hospitals in:
- Technical evaluation
- Site planning
- Vendor comparison
- Installation coordination
- Regulatory guidance
Selection based on hospital workflow and expansion plans ensures long-term sustainability.
Step-by-Step Buying Framework
- Define clinical roadmap (5–10 years)
- Assess patient demographics
- Finalize budget range
- Compare detector and imaging specs
- Verify AERB compliance readiness
- Review service contract terms
- Negotiate training and software upgrades
“A Cath lab purchase must align with future expansion plans, not just current needs.”
Frequently Asked Questions (FAQ)
₹1 crore to ₹8 crore depending on brand, configuration, and detector size.
Minimum 800–1,000 sq ft with radiation shielding and control room.
Both offer advanced imaging and dose management. Final selection depends on workflow preference, structural heart focus, and service availability.
Yes. No Cath lab in India can operate without clearance from the Atomic Energy Regulatory Board.
3–5 months including civil modification, shielding, delivery, and calibration.
Conclusion
Selecting a Cath lab machine is a long-term strategic decision that affects clinical precision, radiation safety, regulatory compliance, and financial sustainability. Hospitals must align system capabilities with projected procedure volume, infrastructure readiness, and expansion goals. Careful evaluation of imaging technology, dose management features, service support, and compliance.
Leading platforms from Philips and Siemens Healthineers offer advanced solutions, but final selection must depend on clinical alignment rather than brand preference alone. SASHA HEALTH CARE assists hospitals with structured technical assessment, planning, and implementation support to ensure informed and sustainable investment decisions.
“A well-planned Cath lab delivers measurable clinical outcomes and long-term institutional value.”